Plasma Trivia 🧩
Under FDA regulations, how frequently is a source plasma donor permitted to donate?
A) Once per week, no more than four times per month
B) Once every two days, no more than twice in a seven-day period
C) Once every three days, no more than twice in a ten-day period
D) Once per week, with a mandatory 48-hour rest between donations
Answer below.
TODAY’S SPONSOR
Daily news for curious minds.
Be the smartest person in the room. 1440 navigates 100+ sources to deliver a comprehensive, unbiased news roundup — politics, business, culture, and more — in a quick, 5-minute read. Completely free, completely factual.
PLASMA MARKET WATCH
THE WEEK IN PLASMA
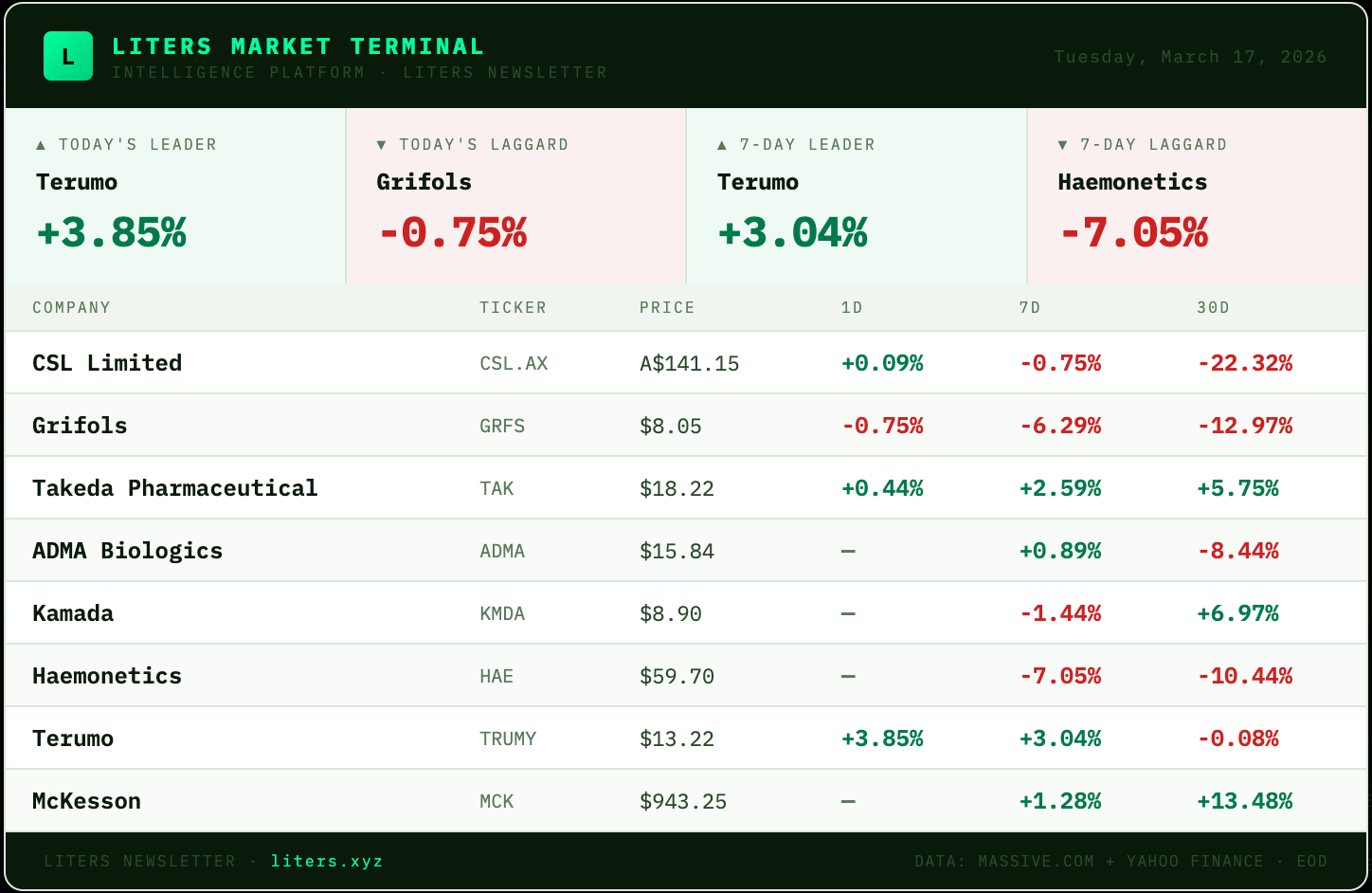
Haemonetics fell 7.05% to $59.70, making it the worst performer in the core plasma names. The company earlier this week repaid $300 million in convertible notes on March 2 and presented at the Raymond James conference, but no specific operational catalyst explains the decline. Takeda gained 2.59% despite announcing organizational changes that included moving Giles Platford from heading its Plasma-Derived Therapies Business Unit to lead a new International Business Unit, with the company still recruiting a plasma business successor.
WHAT IT MEANS
CSL's 81% profit drop and 4% revenue decline in H1 2026 reported in February continues to weigh on the plasma manufacturing leader, which fell another 0.75% this week. The performance divergence between equipment providers and plasma manufacturers is stark: Terumo rose 3.04% while the pure-play manufacturers struggled. This suggests investors are differentiating between companies dependent on plasma collection volumes versus those selling equipment into a recovering procedure environment.
WHAT TO WATCH
Takeda is actively recruiting a new head for its Plasma-Derived Therapies Business Unit after Platford's reassignment, signaling potential strategic shifts ahead for one of the sector's major participants. The leadership transition comes as the company focuses on three core therapeutic areas where plasma represents roughly 40% of exposure.
Liters weekly market analysis is brought to you by the Liters Market Terminal available to Liters+ members; upgrade for full terminal access.
MAIN STORY

For decades, the global plasma industry has rested on a single structural fact: the United States collects approximately 65% of the world's source plasma because it pays its donors, and almost no one else does at scale.
Europe consumes more plasma-derived medicines than any other region but produces a fraction of what it needs, importing roughly 40% of its plasma requirements from the U.S. The dependency is so entrenched it has become background noise in supply chain discussions.
The conventional wisdom held that changing this required either legalizing compensated donation across Europe, which is politically radioactive, or waiting for a technological substitute that hasn't arrived. Neither was happening soon.
On December 11, 2025, that structural assumption developed a crack.
On that date, the European Medicines Agency (EMA) issued a Certificate of Compliance for the entire value chain of Grifols Egypt for Plasma Derivatives (GEPD), a joint venture between Grifols S.A. (NASDAQ: GRFS) and Egypt's National Service Projects Organization. The certification covers every step of the chain: donor medical assessment, plasmapheresis collection, testing, plasma processing, and the supply of finished medicines to patients.
Egypt is now the first country in Africa and the Middle East with a fully EMA-certified, end-to-end plasma ecosystem. More importantly, it is the first compensated-donor plasma platform outside the United States and the four EU countries that permit donor compensation, specifically Germany, Austria, Czechia, and Hungary, to receive that certification.
That last detail is the one the industry should be paying attention to.
Five Years, €280 Million, and a Proof of Concept
The GEPD joint venture was signed in 2020. Under its terms, Egypt's NSPO holds 51% and Grifols 49%. The combined investment stands at €280 million.
As of the EMA certification in December 2025, GEPD operates 16 donation centers across Egypt, with four more planned for 2026, bringing the network to the 20-center target set in the original agreement.
The fractionation plant tells the forward story. Phase 1 of GEPD's manufacturing facility, located on a 105,000-square-meter site in Egypt's New Administrative Capital, comes online in 2026. When complete, it will have fractionation capacity of up to 1 million liters of plasma annually, with the option to double output to 2 million liters. The 20 donation centers carry an initial collection capacity of approximately 600,000 liters per year.
The sequence matters. Egypt first achieved self-sufficiency in immunoglobulins (IVIG), albumin, and clotting factors for its own population, becoming only the sixth country in the world to do so. Then came the EMA certification. The EMA certification is the export trigger.
Once national needs are met, surplus Egyptian plasma-derived medicines can flow into European markets. Grifols said as much in its FY2025 results, explicitly positioning Egypt as a platform for "reducing reliance on costlier U.S. plasma" and targeting EMEA region supply expansion.
The Regulation That Made It Easier
Here is the part of the story that hasn't been told.
The EU passed its new Substances of Human Origin regulation, known as SoHO, formally Regulation EU 2024/1938, in May 2024, partly in response to the plasma supply crisis. The EMA's own shortage notification reported that immunoglobulin shortfalls were affecting 14 European countries in 2024, with disruptions expected to continue at least through June 2026. The SoHO Regulation explicitly identifies plasma as a "critical" substance and urges Member States to increase collection capacity.
But the SoHO Regulation preserved the Voluntary Unpaid Donation (VUD) principle as the governing framework for European collection. It formally discouraged the compensated model that produces the volume Europe actually needs.
The evidence on this is not subtle. The four EU countries that currently permit compensation already collect significantly more plasma per capita than countries that don't. The PPTA, the Plasma Protein Therapeutics Association and the industry's primary trade body, called the regulation a "missed opportunity" when the European Parliament voted on it in 2023, arguing that restrictive amendments on compensation would entrench rather than solve the supply deficit.
The result is a structural paradox. Europe's new framework for plasma security acknowledges the shortage and urges more collection, but declines to adopt the economic mechanism that would reliably produce it.
Into that gap, Europe needing more plasma and unwilling to pay for it domestically at scale, Grifols has inserted an EMA-certified, non-European, compensated alternative. The regulation designed to reduce third-country dependency has made Grifols' third-country supply play more commercially viable, not less.
A Playbook, Not a One-Off
What makes the Egypt development strategically significant, beyond its immediate value to Grifols, is its replicability.
The template is now proven: build to EMA standards from inception, pursue PPTA certification for collection quality, obtain EMA inspection and compliance sign-off, achieve national self-sufficiency first, then unlock export. GEPD is the first-ever Egyptian source member of the PPTA, meaning the industry's own quality credentialing body has independently validated the model.
Grifols is already running a version of the same playbook in a second geography. Under a 15-year agreement with Canadian Blood Services signed in 2022, Grifols is building a 600,000-liter/year collection network in Canada, with 17 centers contracted and a Montreal fractionation plant targeting full operational capacity by end of 2027. Canada's domestic immunoglobulin sufficiency has climbed from approximately 15% to 31% since the agreement began.
Egypt and Canada are structurally different. Different regulatory environments, different donor compensation models, different national health partnerships. But the underlying logic is identical: identify a market with acute plasma dependency on U.S. supply, propose a sovereign self-sufficiency model, embed Grifols as the technology and operations partner, and build the infrastructure that creates long-term, locked-in plasma flow.
In Canada, that flow is contractually dedicated to Canadian patients for 15 years. In Egypt, once domestic needs are met, the surplus flows toward European markets, where the shortage is real, ongoing, and structurally unsolved.
What Everyone Else Should Be Asking
The question the industry should now be asking is not whether Grifols' Egypt model works. The EMA said it does.
The question is what it means for every other operator still running a strategy built around U.S. collection dominance as a permanent condition.
Grifols now holds something its competitors don't: a certified, cost-advantaged, non-U.S. supply vector into the world's largest plasma-consuming market, with a second one under construction in Canada.
Every major Western plasma operator has expanded internationally, but only within already-established frameworks. CSL collects in Germany, Hungary, and China. Takeda BioLife operates in Austria, Czech Republic, and Hungary. Octapharma and Kedrion collect in Germany and Czech Republic respectively. Every one of those geographies is either a U.S.-equivalent regulatory environment or an existing EU compensation-permitted market.
Not one has built a new-market platform outside those established frameworks and taken it through EMA certification for European distribution.
The sovereign self-sufficiency model is spreading beyond the Western incumbents. SK Plasma, the South Korean fractionator, is building a comparable joint venture with Indonesia's sovereign wealth fund, a 600,000-liter/year fractionation plant now more than 98% complete, with first plasma already shipped for toll processing in March 2025. But SK Plasma's Indonesia operation targets domestic Indonesian supply, not European export, and carries no EMA certification.
Grifols is the only operator that has executed the full sequence: new market, new regulatory territory, EMA certification, European export mandate.
If European procurement agencies begin actively diversifying away from U.S.-sourced plasma, which the SoHO Regulation's own language suggests they should, Grifols is currently the only large-scale operator with a certified alternative ready to supply them.
That is a durable competitive position.
TOP 5 HEADLINES
Here's your plasma news roundup for the week of March 11–17, 2026 🩸
Grifols Reports €7.5B Revenue, More Than Doubles Net Profit for FY 2025 — Feb. 26 | 🌍 Spain | Grifols reported full-year 2025 revenues of €7,524 million, representing 7% growth, driven largely by its immunoglobulin franchise. Net profit more than doubled to €402 million from €157 million in 2024 — a 156% increase — with the company pointing to higher operating margins and lower financial costs.
CSL Breaks Ground on $1.5B Illinois Immunoglobulin Plant Expansion — Mar. 9 | 🌍 USA | CSL Behring broke ground on a $1.5 billion expansion of its manufacturing facility in Kankakee, Illinois, aimed at substantially increasing its capacity to produce plasma-derived therapies for people with rare and serious diseases like hemophilia and immune deficiencies. The expansion will incorporate CSL's Horizon 2 manufacturing process — a yield-enhancing technology designed to produce greater quantities of immunoglobulin from the same base amount of plasma — and is expected to come online by 2031. Illinois Governor Pritzker noted this would be the first time CSL's full manufacturing process, from plasma collection to final filling and packing, would be entirely on U.S. soil.
Two Donors Die at Grifols Clinics in Winnipeg; Health Canada Investigates — Mar. 11 | 🌍 Canada | CBC News reported that two people died after giving plasma at Grifols donation centres in Winnipeg — one in October 2025 and another on January 30, 2026 — with Health Canada confirming it received reports of fatal adverse reactions and conducting on-site inspections at both locations. One of the donors was Rodiyat Alabede, a 22-year-old international student from Nigeria studying at the University of Winnipeg; the deaths took place at two separate Grifols locations. Grifols said an internal review found "no reason to believe" the deaths were linked to plasma donation, but the story quickly escalated into a broader public safety and regulatory debate in Canada.
Grifols Expands AMBAR Alzheimer's Program to New Spanish Cities — Mar. 12 | 🌍 Spain | Grifols renewed its agreement with Ace Alzheimer Center Barcelona to continue developing the AMBAR clinical program — which uses periodic plasma exchange with albumin to slow Alzheimer's progression — and announced new AMBAR Clinic centers opening in Madrid, Mallorca, and Alicante. The AMBAR approach has been shown to slow disease progression by 61% in moderate Alzheimer's patients, and the program has now performed over 5,000 plasma exchanges since it launched in 2004.
Grifols Doubles Revolving Credit Facility to $2 Billion — Mar. 16 | 🌍 Spain | Grifols secured commitments from a global bank syndicate for a new revolving credit facility of approximately $2 billion — more than doubling its previous RCF of $938 million — with the margin dropping from 300 basis points to 200 basis points, and potential for further reduction tied to deleveraging progress. The move signals growing confidence from lenders in Grifols' financial recovery trajectory, coming on the heels of recent credit rating upgrades from both S&P and Moody's.
Trivia Answer 🧩
B. Under FDA regulations, source plasma donors may donate no more than once in a two-day period, and no more than twice in any seven-day period. This frequency is significantly higher than whole blood donation (which is limited to once every 8 weeks), and is made possible by the plasmapheresis process, which returns red blood cells to the donor, allowing plasma to replenish much more quickly than whole blood components.
Disclaimer: Liters is an independent research and analysis publication covering the plasma industry, published by Liters Inc. Liters Inc. also operates Bell Chime Capital, an investment platform focused on plasma-backed assets, so topics covered may occasionally overlap with areas of investment interest. Liters is not promotional and does not solicit capital or provide investment advice.